Capped Bottle and Water Flow | Pressure Switch
The concept of air pressure is studied at various levels. This demo illustrate it in a dramatic way.
Procedure
Make two small holes on the side of the bottle, one near the top (upper hole) and other near the bottom (lower hole). Fill the bottle with water and put a cap on it. Allow the water to fall through the lower hole. Now, close the upper hole with your thumb. Water stops falling. You can make the water fall or stop by closing or opening the upper hole (switch).
Discussion
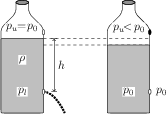
When the upper hole is open, the pressure of air trapped inside the bottle is equal to the atmospheric pressure (say \(p_0\)). The pressure outside the lower hole is also equal to the atmospheric pressure. The pressure inside the bottle at the level of lower hole is atmospheric pressure plus pressure due to water column i.e., \(p_l=p_u+h\rho g=p_0+h\rho g\). Thus, there is pressure difference between inside and outside of the lower hole and water starts flowing out. When the upper hole is closed, if you watch closely, you will notice that the water does not stop falling immediately. The water continues falling for a moment and then stops. Due to this, the volume of air trapped inside the bottle increases and its pressure decreases (Boyle's law). Thus, pressure inside the bottle at the level of lower hole also decreases. When this pressure becomes equal to the atmospheric pressure, the water stops flowing out from the lower hole.
Related
Subscribe to our channel
