Thought provoking problems on modern physics
These problems are from pre-test conducted in teacher's workshops by Prof HC Verma and his team.
Test 1
Source: RESPECT KV MRP Workshop 15th-24th May 2017
- The electric field in a metal is zero in electrostatics. But in Rutherford's experiment, the alpha particle gets deflected showing presence of electric field there. This is because,
- Gold is not a metal but semi metal
- When alpha particle moves in gold, this is not an electrostatic situation.
- The moving alpha particle creates a magnetic field which shifts the electron clouds to create the electric field.
- The electric field inside a metal is zero only in an average sense.
- The alpha particles are deflected from the surface of the gold foil and at the surface of a metal.
- A helium nucleus is stable because the nuclear attractive force dominates over Coulomb repulsion. Why 4 neutrons do not make a stable nucleus where there should be a strong nuclear attractive force but no Coulomb repulsion.
- A gamma decay is most often preceded by either alpha decay or beta decay. This because
- gamma rays are produced by alpha or beta particles
- a gamma decay alone does not conserve momentum conservation
- in alpha or beta decay, the daughter nucleus is prepared in excited state
- in nuclear reactions, the energy released is so large that gamma ray alone cannot take up this energy alone.
- A voltage is applied to a hydrogen discharge tube just enough to produce a faint orange light. Together with this,
- a lots of ultraviolet radiation must be coming
- a lots of infrared radiation must be coming
- a lots of visible light of different wavelengths must be coming.
- In Bohr's model, the angular momentum of the electron in hydrogen atom for $n=1$ orbit is Planck's constant/($2\pi$). But this electron is also called 1s electron having angular momentum $l = 0$. Which of the two is more realistic?
- A photo cell is exposed to light of frequency n1 in one experiment and of a higher frequency n2 in another experiment. The intensity of light is kept same in the two cases.
- In which case, the number of photons falling on the cathode is more? Or it is same in the two cases?
- Sketch the photocurrent I vs anode voltage for the two cases.
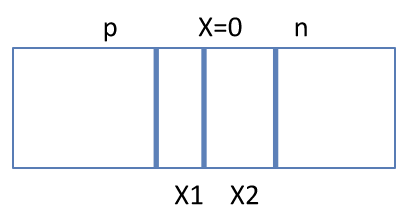
- A pn junction is schematically shown in the figure. The depletion region extends for a width x1 on the p-side and for a width x2 on the n-side.
- x1 must be equal to x2.
- p-type extends from the left end to x = -x1 and n-type extends from x=x2 to the right end.
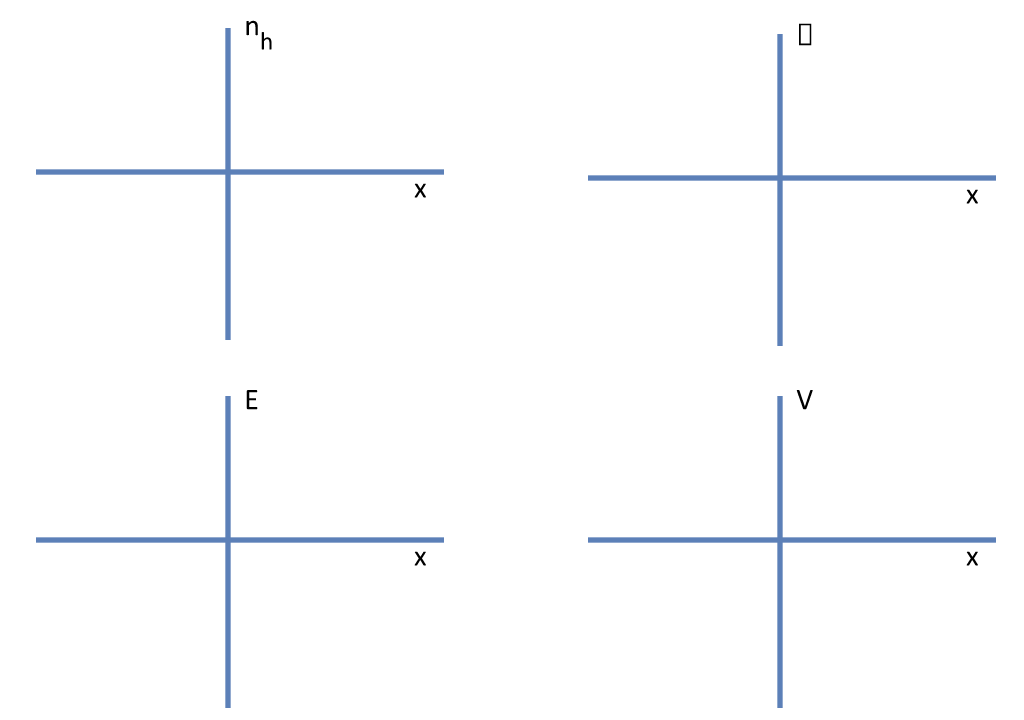
- Sketch the graph of the following as a function of x. (i) $n_h$ (number of holes per unit volume), (ii) charge density $\rho$, (iii) electric field $E$, and (iv) potential (taking V = 0 at x = 0).